


BIOMOR® Curcumin is a clinically-proven and patented combination of curcuminoids and essential oils—with no additives—which results in up to 800% greater curcumin absorption in humans—and it's faster-acting and longer-lasting compared to other 95% standardized curcumin extracts. (8,9,23)
BIOMOR® Curcumin has been extensively tested in clinical trials at academic institutions worldwide.
BIOMOR® Curcumin is composed of a patented combination of curcuminoids and essential oils—with no additives.
Even though many herbs and medicinal plants contain various biologically active compounds that may trigger side effects and interact with other herbs, supplements or medications, curcumin (without piperine) is considered very safe. (1)
BIOMOR® Curcumin has been tested for toxicity in peer-reviewed safety studies. In all investigations, BIOMOR® Curcumin was found safe and nontoxic in high doses. There were no significant changes in hematological and biochemical parameters. Interestingly, researchers found high doses of BIOMOR® Curcumin caused a healthy decrease in serum cholesterol. (10)
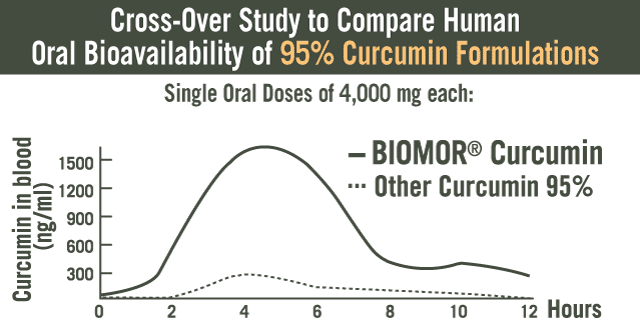
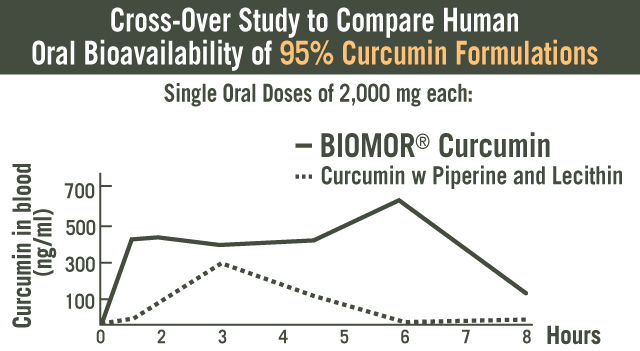
Clinical study evaluated the safety and effectiveness of BIOMOR® Curcumin alone, and in combination with an NSAID. Forty-five subjects were randomized into three groups with subjects receiving BIOMOR® Curcumin (500 mg) and NSAID (50 mg) alone or in combination. The primary and secondary endpoints were reduction in (DAS) 28 Score and ACR score, assessments used by clinicians that measures various joint-related parameters. Subjects in all three treatment groups showed statistically significant changes in their DAS scores. Interestingly, the BIOMOR® Curcumin group showed the highest percentage of improvement in overall DAS and ACR scores (ACR 20, 50 and 70) and these scores were significantly better than the subjects in the NSAID group. More importantly, BIOMOR® Curcumin was found to safe and without adverse events. (29)
The safety and efficacy of BIOMOR® Curcumin and directly compared with a selective COX-2 inhibitor. Treatment was well tolerated and did not produce any adverse effect in subjects, as judged by the vital signs, hemogram, liver and renal function tests. 500 mg administered twice a day, was more successful than administration of the COX-2 inhibitor (100 mg twice a day) for symptom scoring and clinical examination. BIOMOR® Curcumin was found to be safe and no dose-related toxicity was found. (28)
Low-grade inflammation has been associated with obesity-associated disorders. The putative therapeutic properties of citrus and Curcumin polyphenols could be associated with their anti-inflammatory properties. Two diets supplemented either with hesperidin (0•05 %) and naringin (0•1 %) from citrus extract or with BIOMOR® Curcumin from Curcuma longa (0•09 %) were fed to eight obese cats for two 8-week periods (cross-over study design) while maintaining animals in an obese state. Plasma acute-phase protein (APP; a1-acid glycoprotein (AGP), serum amyloid A and ptoglobin) levels were assessed before and at the end of each test period. TNFa, IL-1b, IL-2, IL-4, IL-5, IL-10, IL-12, IL-18, transforming growth factor-b, interferon (IFN)-g mRNA levels were determined in peripheral blood mononuclear cells (PBMC) by real-time PCR. Compared with pre-study values, supplementation with citrus polyphenols resulted in lower plasma AGP and haptoglobin concentrations, while that with curcumin resulted in lower plasma AGP concentration. There were no differences between the supplementations. TNF-a, IL-1b, IL-4, IL-5, IL-10, IL-12, IL-18, transforming growth factor-b, mRNA levels remained unaffected by either dietary supplementation. In contrast, IFN-g and IL-2 mRNA levels were lower at the end of the citrus and the curcumin supplementation, respectively. There were no differences between the supplementations. The present study results show a slight effect of citrus and BIOMOR® Curcumin supplementation on inflammatory markers expressed by PBMC, and a decreased concentration of APP, which are mainly expressed by the liver. This would confirm that hesperidin and naringin or BIOMOR® Curcumin have beneficial effects, targeted in the liver and could improve the obesity-related inflammatory state. (35)
Some previous in vitro and in vivo studies have proposed a radio-protective role for curcumin, the yellow pigment of turmeric. The purpose of this investigation was to assess the radio-protective effects of BIOMOR® Curcumin supplementation in subjects undergoing external beam radio-therapy (EBRT) were randomly assigned to curcumin group, taking 3 g/d curcumin (6 × 500 mg capsules of BIOMOR® Curcumin n=20), or placebo group (n=20). Quality of life was assessed by the quality of life questionnaire (QLQ-PR25). Analysis of covariance was used to compare radiotherapy related symptoms between groups following the intervention, adjusted for baseline symptoms. No differences in urinary symptoms, bowel symptoms, treatment related symptoms and sexual activity were observed between the curcumin and placebo groups before the intervention. The change in urinary symptoms across the 20-week period differed significantly between groups (p=0.011) and subjects in the curcumin group experienced much milder urinary symptoms compared with the placebo group. Results demonstrate BIOMOR® Curcumin can confer radio-protective effect in through reducing the severity of radiotherapy related urinary symptoms. (30)
Thirty-four subjects were eligible for this double-blind, placebo-controlled, randomized, 6-month trial if they were 50 years old or older, ethnic Chinese in Hong Kong, had progressive decline in memory and cognitive function for 6 months, and had met NINCDS-ADRDA criteria. Subjects were randomized in 3 Groups each taking 4 grams of placebo, 1 gram of BIOMOR® Curcumin and 4 grams of Conventional Curcumin Extract. At 0, 1 and 6 months plasma was taken to measure antioxidants and serum to monitor AB and liver and kidney functions. Over 1 month, Vitamin E levels in plasma were significantly increased in BIOMOR® Curcumin group compared to others. Moreover, subjects taking BIOMOR® Curcumin had greater levels of curcuminoids than the other. Serum beta-Amyloids tended to rise possibly reflecting an ability of Curcumin to disaggregate beta-Amyloids deposits in the brain, releasing them for circulation and disposal. Where there were few adverse events on Conventional Curcumin group, there were no side effects reported in BIOMOR® Curcumin group. (34)
In the present study, the mood-modulating activity of BIOMOR® Curcumin and its combination with SSRIs was studied in acute model (three doses 24, 5 and 1 h before test) of forced swimming test (FST) in glass jar and tail suspension test (TST) in mice and in chronic model (14 day study) of FST with water wheel in rats. All the tests were carried out in the following seven groups (n = 6 in each group), administered orally (doses for mice): Both the acute model of FST and TST, and the chronic model of FST with water wheel showed significant activity of curcumin in 100 mg/kg dose as compared to vehicle control (p < 0.05). The effect of curcumin (100 mg/kg) was similar to that of SSRIs (p > 0.05) but its addition to SSRIs did not improve their activity (p >0.05). Curcumin increased both the swimming and climbing behavior in FST, thus its mood-modulating activity could be due to an increase in serotonin, norepinephrine and dopamine levels in the brain. Results show BIOMOR® Curcumin can be a useful mood-modulator especially in cases which respond to drugs having mixed effects on serotonin and catecholamines levels in the brain. (31)
A randomized comparative human clinical study was done to evaluate the effect of BIOMOR® Curcumin on mood. Sixty subjects were randomized to receive 20 mg/d of a SSRI (n=20), 500 mg BIOMOR® Curcumin (n=20), and SSRI plusBIOMOR® Curcumin (n=20) for 6 weeks. The 17-item Hamilton Rating Scale (HAMD-17) was the primary efficacy measurement. CGI-S was the secondary efficacy measure. Overall compliance by the study participants was excellent. The proportion of responders as measured by the HAM-D17 scale was higher in the combination group (77.8%) than in the SSRI (64.7%) and the curcumin (62.5%) groups; however, these data were not statistically significant (P = 0.58). Interestingly, the mean change in HAM-D17 score at the end of six weeks was comparable in all three groups (P = 0.77). (32)
Several studies have demonstrated the healthy mood support effects of curcumin. In a randomised, double-blind, placebo-controlled study, 123 individuals were allocated to one of four treatment conditions. Results show that differing doses of BIOMOR® Curcumin is effective in supporting healthy mood function. (33)

Both my wife and I are taking Biomor Curcumin. It has been very effective for both of us. We have tried many brands of curcumin, including one that appears very similar except the other is manufactured using stearates. I felt a very significant difference between Biomor Curcumin and the other brands. I also found Biomor Curcumin is less expensive because I take less, with better results. I'm very happy with Biomor Curcumin. —Mr. J.S.*
I judge by my knees! My brother gave me a bottle of Biomor Curcumin and it works much better for me than the other brands of curcumin. —Mr. M.V.*
I have been taking your BioMor Curcumin for about 6-8 months now. I will always take it. I have a bad back and it helps alleviate swelling and pain from that but I have done some reading about the longer term benefits of taking it and am so glad to be a customer... Thank you. —Mr. Ed K.*
My husband is a lumber grader and so he uses his shoulders and arms a lot. He has been taking 2 BioMor Curcumin and 2 Flavay Plus per day and he noticed how his shoulders and arms are feeling better. Even his psoriasis is better. Thank you for helping us with something that helps my husband so much! —Ms. D.E.*
Both my wife and I are taking Biomor Curcumin. It has been very effective for both of us. We have tried many brands of curcumin, including one that appears very similar except the other is manufactured using stearates. I felt a very significant difference between Biomor Curcumin and the other brands. I also found Biomor Curcumin is less expensive because I take less, with better results. I'm very happy with Biomor Curcumin. —Mr. J.S.*
I don’t have to take aspirin anymore, this Biomor Curcumin is really working for me. But when I stop taking it, my pain comes right back. —Ms. Lynda C.*
Now that I’m taking 4 BioMor Curcumin every day my pain is gone. Only about once a month, I’ll take an antiiflammatory for pain in my knees. I’m feeling so much better. —Ms. Y.L.*
Thank God I found your Biomor Curcumin! My nutritionist recommended I take 5,000 mg curcumin per day, so I tried several brands of curcumin. Of one brand with piperine, I took 8 per day of 580 mg per capsule (4,440 mg per day). Of another brand, also with piperine, I took 6 per day of 875 mg per capsule (5,250 mg per day). Both of the curcumin products with piperine caused me to feel extremely exhausted, to the point where I couldn't do anything but rest. It seems the piperine reacted with my medications. So, I'm so glad to have to have found Biomor Curcumin. I began feeling good with only one 500 mg capsule before bedtime, and I will begin to take another in the morning (1,000 mg per day). I feel great taking Biomor Curcumin. —Mr. N. Smith*
I’ve had good luck with Biomor Curcumin, my arthritic finger cleared up. —Mr. Daniel F.*
I want to tell your company that your Biomor Curcumin is doing wonderful things for my wife. We've tried various curcumin products and this one is the best. —Mr. R.B.*
It (Biomor Curcumin) has been working GREAT for my arthritic knees!!! I was going to have additional shots but decided to try these instead. AMAZING RESULTS!!!! —Mr. B.C.*
My daughter takes Biomor Curcumin for boils and I want to tell you it really helps. —Ms. T.C.*
I judge by my knees! My brother gave me a bottle of Biomor Curcumin and it works much better for me than the other brands of curcumin. —Mr. M.V.*
I'm getting relief from arthritis symptoms in my right knee (taking both Flavay and Biomor Curcumin). Sitting for long periods or getting out of my car was very painful for those first 5-10 steps, but no longer. The swelling in my knee has also subsided and the range of motion has come back... I'm definitely much better and in less pain than before. Another benefit I also noticed is that dry skin in both of my lower legs/ankle area has disappeared. —Mr. B.M.*
Thank you for Biomor Curcumin. I have a muscle disease which affords me little strength, and severe osteoarthritis. I cannot take NSAIDs or steroids due to severe diverticulosis. Biomor Curcumin gives me the flexibility to be able to move about freely. I was about to enter a nursing facility and was introduced to your fine product. —Mr. Kenneth W.*
I am not one to review products, but i can't deny some of the benefits i've experienced using both your curcumin and flavay products. I've been taking both for a few months now, and the two major benefits i've noticed are 1) joint strength and mobility (for workouts) as i am lifting much heavier weights without injury 2) I am a paroxsymal Afib sufferer, and my episodes have gone from 2-3 per week down to zero (most weeks). This can't just be a coincidence. I think there is definitely something in your formulas that stop inflammation and get your body in balance and healing itself. —Mr. B.V.W.*
I am your best Fan...Go BIOMOR...Ra Ra Ra... 😊 😊 —Mr. George D.*
I can't believe what this stuff (BioMor Curcumin) has done for me! I just can't believe it! I was in three car wrecks and so I could hardly move and I was so stiff. But after about 3 weeks jumped out of bed! —Mr. Mike T.*
I've been taking one BioMor Curcumin a day for a long time. It does help. It works well, that's all I'll say. —Mr. John R.*
My husband's hands were hurting so bad but after Biomor Curcumin his hands feel aren't hurting! —Ms. Wanda M.*
Thank you so much for your quick response. The Biomor Curcumin has helped me so much with my Rheumatoid Arthritis, Osteoarthritis and M.S. When I don’t take the Biomor you can see the swelling worsen in my joints, it has helped me so much since I started taking them, and will now add an extra pill or two during the times my inflammation is at it’s worse. I will also continue to advise others to research, and purchase Biomor through Healthy Source. —Ms. B.H.*
I bought BioMor Curcumin for my husband because he had hip surgery years ago and he has a lot of pain on certain days and he says it totally works for him. He doesn’t want to take any more pain killers and this is working for him. —Ms. S. Hill*
I’ve tried other brands and you cannot compare, not even close. I play golf and no more pain when taking BioMor Curcumin! You guys are the best! —Mr. Jack K.*
I’m so thrilled with BioMor Curcumin. There’s no question in my mind this is a miracle. I have RA and after taking BioMor Curcumin my legs and ankles are stronger and I feel so much better. I’m very grateful. (I tried other and much more expensive curcumin products and there is no comparison.) —Ms. J.B.*